dstroy
Well-Known Member
I got the dimmer circuit fixed, and I'll share it with you so that anyone can build it. It was a little more involved than I thought it would be initially but all the parts are really cheap.
I added two switches and two 10kohm potentiometers to the front panel of the control box. The switches are an easy way to switch the different grow areas from veg to flower without having to upload a different program to the Arduino when it's time to flip, all they do is tell the Arduino if they are open or closed and the program switches the photoperiods to 12 or 18 hours. The 10kohm potentiometers allow me to manually dim the lights.
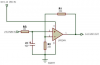
So instead of using transistors (PWM, pulse width modulation) I ended up using an LM324N op amp (additive voltage, 0-10v). It has 4 channels available (perfect!) so that each meanwell driver has its' own dimming circuit.
I'm not sure why the PWM dimming circuit didn't work, but probably had something to do with there only being two channels out and I have 4 drivers so I had to wire them in parallel.
But this one is working great!
Each individual circuit looks like this:
and it looks like this when it's all put together

Don't mind my messy protoboard, I'm not the best at point to point wiring.
I added some more stuff to my serial monitor:
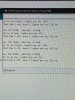
(all of this stuff gets datalogged, at 5 second intervals)
and the wiring of the switches/pots


The only other thing that I've done is to add large (10A) flyback diodes for the solenoid circuits.
I added two switches and two 10kohm potentiometers to the front panel of the control box. The switches are an easy way to switch the different grow areas from veg to flower without having to upload a different program to the Arduino when it's time to flip, all they do is tell the Arduino if they are open or closed and the program switches the photoperiods to 12 or 18 hours. The 10kohm potentiometers allow me to manually dim the lights.
So instead of using transistors (PWM, pulse width modulation) I ended up using an LM324N op amp (additive voltage, 0-10v). It has 4 channels available (perfect!) so that each meanwell driver has its' own dimming circuit.
I'm not sure why the PWM dimming circuit didn't work, but probably had something to do with there only being two channels out and I have 4 drivers so I had to wire them in parallel.
But this one is working great!
Each individual circuit looks like this:

and it looks like this when it's all put together

Don't mind my messy protoboard, I'm not the best at point to point wiring.
I added some more stuff to my serial monitor:

(all of this stuff gets datalogged, at 5 second intervals)
and the wiring of the switches/pots


The only other thing that I've done is to add large (10A) flyback diodes for the solenoid circuits.
























